Research

Systemic acquired resistance (SAR) is a form of induced resistance in plants; it manifests as priming, resulting in faster and/or stronger defense responses in systemic tissues of plants undergoing a local pathogen infection.
As part of the DFG-funded SFB924 (Molecular Mechanisms of Yield and Yield Stability in Plants), we identified key players in SAR signaling. These include volatile organic compounds such as monoterpenes in the model plant Arabidopsis thaliana and the C9-aldehyde nonanal in the cereal crop barley (Brambilla et al., 2022, 2023, J Exp Bot; Riedlmeier et al., 2017, Plant Cell; Wenig et al., 2019, Nat Commun). Exposure of receiver plants (R) to the volatile emissions of infected senders (S) enhances the immunity of the receiver plants to a subsequent pathogen infection.
https://www.sfb924.wzw.tum.de/overview-sfb924/
In Arabidopsis, we further identified the SAR signaling protein LEGUME LECTIN-like PROTEIN1 (LLP1) which promotes SAR downstream of volatile monoterpenes (Breitenbach et al., 2014, Plant Physiol; Wenig et al., 2019, Nat Commun). In ongoing work, we aim to unravel the molecular mechanism driving SAR in response to the activation of LLP1 and its close homologues LLP2 and LLP3. In doing so, we want to identify new small molecules that might be used in future, durable crop protection schemes.

Further work aiming at enhancing plant immunity through priming is concentrated in the project HortiPrimed - Etablierung von „Priming“ an Tomaten-Jungpflanzen als Pflanzenschutzverfahren im Gartenbau. Funded by BMEL-BÖL and together with partners in Jülich, Geisenheim, Munich, and Bonn, we aim to prime defense responses in the horticultural crop tomato by applying abiotic stress treatments at the seedling stage. In our part of the project, we optimize priming against a broad range of pathogens and further aim to understand the molecular process underlying the priming response.
In a project that is part of the DFG-funded priority program 2125 – Deconstruction and Reconstruction of the Plant Microbiota – we further aim to understand the interplay between plants and their microbiome. Changes in the composition of the microbial populations on plant’s roots and leaves influence immunity and in turn respond to changes in the immune status of the plant host. In our project, we aim to understand this interplay between the plant microbiome and induced [disease] resistance.
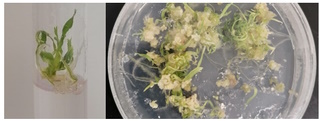
Throughout our research we exploit CRISPR-Cas9-mediated genome editing to expedite the characterization of molecular mechanisms underlying priming. To this end, we aim to establish a gene editing and transformation pipeline for the cereal crops barley and wheat. As a staple crop, wheat plays an important role in human nutrition. Nevertheless, it’s genome has remained poorly understood. By implementing genome editing in wheat, we aim to contribute to a better understanding of innate immune responses in wheat at the molecular and genetic level; in turn, this will allow us to better predict wheat responses to environmental stress and disease and to provide direction to future crop protection strategies to maintain or improve yield quantity and quality.

Funding


